Common Types of Corrosion
GeneralAccelerated electrochemical corrosion that occurs when two dissimilar metals in electrical contact are made part of a circuit completed by pool or film of electrolyte or corrosive media leading to a current flow and ensuing corrosion
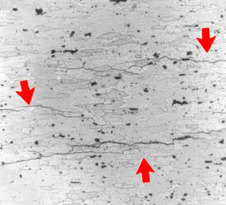
intergranularLocalized attack at grain boundaries (Cu,Cr, Ni, Al, Mg, Zn, SS) when improperly heat-treated or welded.
DealloyingSelective leaching of an element in an alloy. A common example is the dezincification of unstabilized brass, whereby a weakened, porous copper structure is produced.
|
PittingLocalized electro-chemical corrosion at the surface leading to pits. Considered to be a dangerous form of corrosion because it is difficult to detect, predict, and design against.
ErosionAbrasive or viscous fluid which constantly exposes fresh surface material to corrosive material
Hydrogen EmbrittlementGroup of processes in which Hydrogen gas leads to degradation of it's materials and its properties.
|
CreviceAccelerated corrosion localized with in crevices, cracks, or joints where small volume regions of stagnant solution trapped in contact with corroding metal
CavitationCertain bubbles in a fluid collapse due pressure difference along materials surfaces leading to material removal
. ENVIRONMENTAL ASSISTEDCorrosion resulting from living organisms such as MIC or Fungi
|